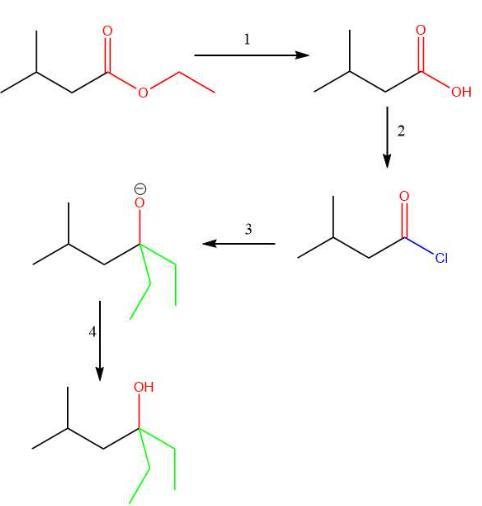
what is an appropriate stepwise synrthesis for the folowing synthesis that uses ethyl 3-methylbutanoate and using any other eagensts necessary
Answers
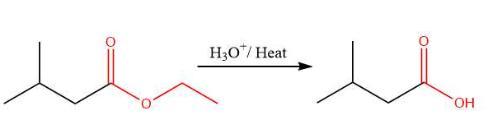
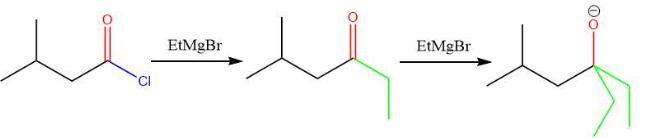
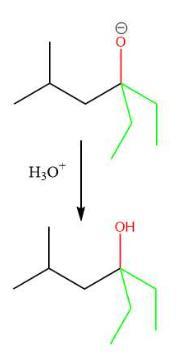
The appropriate stepwise synthesis that uses ethyl 3-methylbutanoate as the only source of carbon is H₃O⁺/heat; 2. SOCI₂; 3. 2 equiv. CH₃CH₂MgBr; 4.H₂O, option B.
As the ester of ethyl alcohol and isovaleric acid, ethyl isovalerate is an organic chemical. It is used as a food additive and in perfumery. It has a fruity aroma.
The group of organic substances known as fatty acid esters includes ethyl isovalerate, also known as ethyl isopentanoate. These are fatty acid carboxylic ester derivatives. According to a survey of the literature, a sizable number of publications have been written about ethyl isovalerate.
To convert the substrate mentioned to the product, we need to see the framework of the following reaction.
The framework of the given conversion is given below,
Learn more about Stepwise synthesis:
https://brainly.com/question/30134818
#SPJ4
Related Questions
the volume of a balloon containing an ideal gas is 3.78 l at 1.05 atm pressure. what would the volume be at 2.75 atm with constant temperature and molar amount? view available hint(s)for part a the volume of a balloon containing an ideal gas is 3.78 l at 1.05 atm pressure. what would the volume be at 2.75 atm with constant temperature and molar amount? 9.90 l 1.44 l 0.764 l 10.9 l
Answers
The volume of the balloon at 2.75 atm pressure with constant temperature and the molar amount would be approximately 1.44 L.
Let's understand this in detail:
We'll use Boyle's Law to solve this question, which states that the product of the pressure and volume of an ideal gas is constant when the temperature and molar amount remains constant.
The formula for Boyle's Law is P1V1 = P2V2, where P1 and V1 are the initial pressure and volume, and P2 and V2 are the final pressure and volume.
Initial volume (V1) = 3.78 L
Initial pressure (P1) = 1.05 atm
Final pressure (P2) = 2.75 atm
Constant temperature and molar amount
To find the final volume (V2), rearrange the formula:
V2 = (P1V1) / P2
Plug in the given values:
V2 = (1.05 atm * 3.78 L) / 2.75 atm
V2 ≈ 1.44 L
So, the volume of the balloon at 2.75 atm pressure with constant temperature and the molar amount would be approximately 1.44 L.
Learn more about Boyle's law: What is Boyle’s law? https://brainly.com/question/1696010
#SPJ11
The volume of the balloon containing the ideal gas would be 1.44 L at 2.75 atm pressure with constant temperature and molar amount.
We can use the ideal gas law to solve this problem: PV = nRT, where P is the pressure, V is the volume, n is the molar amount, R is the gas constant, and T is the temperature. Since we are keeping the temperature and molar amount constant, we can simplify the equation to PV = k, where k is a constant.
Using the initial conditions, we have:
(1.05 atm)(3.78 L) = k
Solving for k, we get k = 3.969 L*atm.
Now, we can use the same equation with the new pressure to find the new volume:
(2.75 atm)(V) = 3.969 L*atm
Solving for V, we get V = 1.44 L.
To learn more about ideal gas, refer:-
https://brainly.com/question/28257995
#SPJ11
which of the following aqueous solutions has the highest molar concentration of na (aq)?(assume each compound is fully dissolved in water.)group of answer choices3.0m nacl (sodium chloride)3.0m nac2h3o2 (sodium acetate)1.5m na2so4 (sodium sulfate)1.0m na3po4 (sodium phosphate)all of these solutions have the same concentration of na (aq).
Answers
All of these solutions have the same concentration of Na⁺ (aq) at 3.0 moles for molar concentration.
The highest molar concentration of Na⁺ (aq) can be determined by calculating the moles of Na⁺ ions in each solution.
1. Identify the number of sodium ions (Na⁺) in each compound:
- NaCl: 1 Na⁺ ion
- NaC₂H₃O₂: 1 Na⁺ ion
- Na₂SO₄: 2 Na⁺ ions
- Na₃PO₄: 3 Na⁺ ions
2. Calculate the moles of Na⁺ ions in each aqueous solution:
- 3.0 M NaCl: 3.0 M * 1 Na⁺ ion = 3.0 moles of Na⁺ ions
- 3.0 M NaC₂H₃O₂: 3.0 M * 1 Na⁺ ion = 3.0 moles of Na⁺ ions
- 1.5 M Na₂SO₄: 1.5 M * 2 Na⁺ ions = 3.0 moles of Na⁺ ions
- 1.0 M Na₃PO₄: 1.0 M * 3 Na⁺ ions = 3.0 moles of Na⁺ ions
3. Compare the moles of Na⁺ ions in each solution to determine the highest concentration.
All of these solutions have the same concentration of Na⁺ (aq) at 3.0 moles.
Learn more about molar concentration here:
https://brainly.com/question/21841645
#SPJ11
Though all the solutions have the same concentration of Na+ (aq), an aqueous solution of NaCl with 3.0 M has the highest molar concentration among the given solutions.
Explanation: To determine the molar concentration of Na+ (aq) in each solution, we need to consider the stoichiometry of the dissociation of each compound in water.
For sodium chloride (NaCl), it dissociates completely into Na+ and Cl- ions, so the molar concentration of Na+ (aq) is equal to the molar concentration of NaCl. Therefore, the molar concentration of Na+ (aq) in 3.0M NaCl is 3.0M.
For sodium acetate (NaC2H3O2), it dissociates into Na+ and C2H3O2- ions, but in a 1:1 ratio. So, the molar concentration of Na+ (aq) is half of the molar concentration of NaC2H3O2. Therefore, the molar concentration of Na+ (aq) in 3.0M NaC2H3O2 is 1.5M.
For sodium sulfate (Na2SO4), it dissociates into 2 Na+ ions and 1 SO4 2- ion. So, the molar concentration of Na+ (aq) is twice the molar concentration of Na2SO4. Therefore, the molar concentration of Na+ (aq) in 1.5M Na2SO4 is 3.0M.
For sodium phosphate (Na3PO4), it dissociates into 3 Na+ ions and 1 PO4 3- ion. So, the molar concentration of Na+ (aq) is three times the molar concentration of Na3PO4. Therefore, the molar concentration of Na+ (aq) in 1.0M Na3PO4 is 3.0M.
Therefore, the solution with the highest molar concentration of Na+ (aq) is 3.0M NaCl (sodium chloride).
To know more about the molar concentration of solutions:
brainly.com/question/8732513
#SPJ11
the tollen's test is the reaction of aldehydes with silver(i) ions in basic solution to form silver metal and a carboxylate. reaction of 2 silver 1 ions with a generic aldehyde and 3 hydroxide ions to form 2 silver atoms, a generic carboxylate, and 2 water molecules. which species is being oxidized in the reaction? aldehyde which species is being reduced in the reaction? silver(i) ion which species is the visual indicator of a positive test? silver metal
Answers
In Tollen's test, the reaction of aldehydes with silver(i) ions in basic solution results in the formation of silver metal and carboxylate.
Specifically, the reaction involves the oxidation of the aldehyde and the reduction of the silver(i) ion. This can be seen in the reaction of 2 silver 1 ions with a generic aldehyde and 3 hydroxide ions, which produces 2 silver atoms, a generic carboxylate, and 2 water molecules. The species being oxidized in the reaction is the aldehyde, while the species being reduced is the silver(i) ion. The visual indicator of a positive test is the formation of silver metal, which indicates the presence of an aldehyde in the sample.
To learn more about Tollen's test, refer:-
https://brainly.com/question/30892406
#SPJ11
In this Tollen's test, the species being oxidized is the aldehyde (RCHO), while the species being reduced is the silver(I) ion (Ag+). The visual indicator of a positive test is the formation of silver metal (Ag), which appears as a shiny silver mirror on the inner surface of the test tube.
What is Tollen's Test?In the Tollen's test, the reaction involves aldehydes reacting with silver(I) ions in a basic solution to form silver metal and a carboxylate. The generic equation for this reaction is:
2 Ag+ + RCHO + 3 OH- → 2 Ag + RCOO- + 2 H2O
In the Tollen's test, aldehydes react with silver(i) ions in basic solution to form silver metal and a carboxylate. The reaction involves the oxidation of the aldehyde and reduction of the silver(i) ion. Specifically, in the presence of 2 silver(i) ions and 3 hydroxide ions, a generic aldehyde is oxidized to form a generic carboxylate and 2 water molecules, while the silver(i) ions are reduced to form 2 silver atoms. The visual indicator of a positive test is the formation of silver metal, which indicates the presence of an aldehyde. Therefore, in this reaction, the aldehyde species is being oxidized.
To know more about Tollen's Test:
https://brainly.com/question/13833774
#SPJ11
question 6 how do electrons in an atom change energy? electrons can only gain energy by leaving the atom (creating an ion). electrons move between discrete energy levels, or escape the atom if given enough energy. electrons can have any energy below the ionization energy within the atom, or escape if given enough energy. electrons can have any energy within the atom, and cannot be given enough energy to cause them to escape the atom. electrons move between discrete energy levels within the atom, and cannot accept an amount of energy that causes them to escape the atom.
Answers
The electrons cannot have any arbitrary energy within the atom, and they can be given enough energy to escape the atom, forming ions.
Electrons in an atom change energy by moving between discrete energy levels, which are quantized states within the atom. These energy levels are determined by the electron's orbitals and the principal quantum number.
Electrons can gain or lose energy through processes like absorption or emission of photons, respectively. When an electron gains enough energy, it can jump to a higher energy level, or
even escape the atom, resulting in ionization. Conversely, when an electron loses energy, it transitions to a lower energy level, emitting a photon in the process.
To learn more about : electrons
https://brainly.com/question/26084288
#SPJ11
nAt T = 1200º C the reaction: P.(g) + 2P2(8) has an equilibrium constant R, 0.612. Suppose the initial partial pressure of Pris 5.00 atm and that of P, is 2.00 atm. Calculate the re- action quotient, Q. and state whether the reaction proceeds to reactants or products.
Answers
Since Q (0) is less than the equilibrium constant R (0.612), the reaction will proceed in the forward direction, moving towards the formation of more products.
The reaction quotient, Q, is calculated using the formula Q = (PPr)^1 x (PP2)^2, where PPr and PP2 are the partial pressures of Pr and P2, respectively. Plugging in the given values, we get Q = (5.00)^1 x (2.00)^2 = 20.00 atm^2.
To determine the direction of the reaction, we compare the reaction quotient, Q, to the equilibrium constant, K. If Q < K, the reaction proceeds forward to products. If Q > K, the reaction proceeds backward to reactants. And if Q = K, the reaction is at equilibrium.
In this case, the equilibrium constant R = 0.612, which means the reaction strongly favors reactants. Since the reaction quotient Q is much larger than the equilibrium constant (Q > K), the reaction will proceed in the reverse direction towards reactants.
To answer your question, we'll first need to correct the given reaction. Assuming the correct reaction is P(g) + 2P₂(g) ⇌ P₃(g), we can proceed.
Given the initial partial pressures, P(P) = 5.00 atm and P(P₂) = 2.00 atm, and no P₃ is mentioned, so we assume P(P₃) = 0 atm initially.
To calculate the reaction quotient, Q, we'll use the expression: Q = [P₃]/([P] * [P₂]^2). Plugging in the initial values, we get:
Q = (0) / (5.00 * 2.00^2) = 0
Since Q (0) is less than the equilibrium constant R (0.612), the reaction will proceed in the forward direction, moving towards the formation of more products.
Visit here to learn more about equilibrium : https://brainly.com/question/4289021
#SPJ11
To calculate the reaction quotient Q and determine whether the reaction proceeds to reactants or products, we can follow these steps:
1. Write down the balanced chemical equation:
[tex]P (g) + 2 P2 (g) ⇌ 3 P (g)[/tex]
2. Given: T = 1200ºC, K = 0.612, initial partial pressure of P is 5.00 atm, and initial partial pressure of P2 is 2.00 atm.
3. Write down the expression for the reaction quotient, Q:
[tex]Q = [P]^3 / ([P] * [P2]^2)[/tex]
4. Plug in the initial partial pressures:
[tex]Q = (5.00)^3 / (5.00 * (2.00)^2) = 125 / 20 = 6.25[/tex]
Now we can compare Q to the equilibrium constant, K, to determine whether the reaction proceeds to reactants or products.
Since Q > K (6.25 > 0.612), the reaction will proceed towards the reactants to reach equilibrium.
To know more about reaction quotient Q :
https://brainly.com/question/9024475
#SPJ11
In a complete sentence, write down a method you could use to determine if an equation is written in the correct way and balanced
Answers
Option (C) is correct. One should count the atoms of each element on both sides of the chemical equation to make sure they are equal and decide whether the equation is balanced and appropriately constructed.
How should a chemical equation be written? What is it used for?The number of moles of a substance created or consumed during the chemical reaction is indicated by the coefficients next to the entity symbols.
How will you determine whether your answer to the rational equation is accurate?Verify each answer to verify sure the result does not result in the original equation's denominator being equal to zero. a denominator in the original equation can be made zero if a solution can be discovered.
To know more about chemical equation visit:-
https://brainly.com/question/30087623
#SPJ1
Question:
What is a method you could use to determine if an equation is written in the correct way and balanced?
One method to determine if an equation is written in the correct way and balanced is to check that the number and type of atoms are the same on both sides of the equation by using the Law of Conservation of Mass.
A chemical reaction has a Q10 of 3. Which of the following rates characterizes this reaction?
a. a rate of 6 at 20°C and 2 at 30°C
b. a rate of 6 at 30°C and 2 at 20°C
c. a rate of 9 at 20°C and 3 at 30°C
d. a rate of 9 at 40°C and 3 at 20°C
e. a rate of 12 at 10°C and 4 at 20°C
Answers
A chemical reaction has a Q10 of 3 option c. a rate of 9 at 20°C and 3 at 30°C is the rates that characterizes this reaction
The Q10 value is a measure of how much the rate of a chemical reaction changes with a 10°C change in temperature. A Q10 of 3 indicates that the rate of the reaction will increase by a factor of 3 when the temperature is raised by 10°C.
Looking at the answer choices, we can see that option a and b have a Q10 value of 2, which is not the same as the given Q10 value of 3. Option e has a Q10 value of 4, which is also not the same.
Option d has a Q10 value of 3, but the rates given are at 20°C and 40°C, which is not a 10°C change in temperature.
Therefore, the only option that fits the given Q10 value and has rates that are 10°C apart is option c, which has a rate of 9 at 20°C and 3 at 30°C. Therefore, the answer is c.
To learn more about chemical reaction click here
brainly.com/question/29762834
#SPJ11
Option c states that the rate of the reaction is 9 at 20°C and 3 at 30°C. The ratio of rates between 20°C and 30°C is 9/3 = 3, which matches the Q10 value of 3.
c. a rate of 9 at 20°C and 3 at 30°C
The Q10 value is a measure of the temperature sensitivity of a reaction, and it is defined as the factor by which the rate of a reaction changes for every 10-degree Celsius change in temperature. A Q10 value of 3 indicates that the rate of the reaction increases by a factor of 3 for every 10-degree Celsius increase in temperature.
This means that the rate of the chemical reaction is consistent with the temperature sensitivity indicated by the given Q10 value, making option c the correct answer.
Learn more about “ chemical reaction “ visit here;
https://brainly.com/question/29039149
#SPJ4
Fossil fuels are fuels such as coal, oil, and natural gas. They are formed over millions of years from the remains of ancient plants and animals. What is the relationship between fossil fuels and energy?
A. Fossil fuels are a form of chemical energy. B. Fossil fuels are a form of nuclear energy. C. Fossil fuels are a form of mechanical energy. D. Fossil fuels are a form of geothermal energy
Answers
Fossil fuels are a form of chemical energy stored from ancient organic matter and are used as a valuable energy source.
The right response is A. Petroleum products are a type of synthetic energy.Petroleum products are hydrocarbons that contain put away energy from natural matter, which is changed over into fuel through a characteristic interaction that requires a long period of time.
At the point when petroleum products are singed, they discharge the put away compound energy as intensity, which can then be changed over into different types of energy, like electrical energy, mechanical energy, or nuclear power.
This makes petroleum products a significant wellspring of energy for human utilization, especially in transportation and power age.Petroleum products are non-sustainable wellsprings of energy, implying that they will ultimately run out.
Also, the consuming of non-renewable energy sources discharges carbon dioxide and other ozone harming substances into the air, adding to environmental change.
This has prompted expanded interest in sustainable power sources, for example, sunlight based, wind, and hydroelectric power, which don't create ozone harming substance emanations and are not expose to consumption like petroleum products.
To learn more about fossil fuels, refer:
https://brainly.com/question/3514202
#SPJ4
100 POINTS - A sample of crushed rock is found to have 4. 81 x10^21 atoms of gold, how many moles of gold are present in this sample? SHOW WORK INCLUDING FORMULA : THANK YOU
Answers
There are 0.00799 moles of gold present in the sample of crushed rock.
The formula to convert the number of atoms of an element to moles is:
moles = number of atoms / Avogadro's number
where Avogadro's number is approximately 6.022 x 10^23.
Using the given information, we can calculate the number of moles of gold present in the sample:
moles of gold = 4.81 x 10^21 atoms / 6.022 x 10^23 atoms/mol
moles of gold = 0.00799 mol
Note: The answer has been rounded to five significant digits in accordance with the significant figures of the given number of atoms.
To know more about atoms, here
brainly.com/question/30898688
#SPJ4
hydrogen bonding is crucial to the transpiration-cohesion-tension mechanism. true or false
Answers
The given statement "hydrogen bonding is crucial to the transpiration-cohesion-tension mechanism" is true because it enables the cohesive properties of water that allow for efficient water transport in plants.
The transpiration-cohesion-tension mechanism is the process by which water is transported through the xylem tissue of plants from the roots to the leaves. This mechanism relies on the cohesion of water molecules and the tension created by transpiration (the loss of water vapor through the stomata of leaves).
Hydrogen bonding, which is a type of chemical bonding between the hydrogen atom of one molecule and the electronegative atom of another molecule, is crucial to the transpiration-cohesion-tension mechanism. The cohesion of water molecules is due to the presence of hydrogen bonds between adjacent water molecules.
As water molecules evaporate from the surface of leaves during transpiration, they create a negative pressure (tension) that pulls additional water molecules up through the xylem tissue. This process of water transport is only possible due to the strong hydrogen bonds between water molecules, which allow them to stick together and resist the force of gravity.
To learn more about hydrogen bonding, here
https://brainly.com/question/17659933
#SPJ4
A respiratory pigment that requires a relatively low O2 partial pressure for loading has ______ affinity for O2. a) a low b) a high c) no d) a variable.
Answers
A respiratory pigment that requires a relatively low [tex]O_2[/tex] partial pressure for loading has a high affinity for [tex]O_2[/tex]. Thus, the correct answer is an option (a).
Since the respiratory pigment requires low partial pressure of the gas, it has more affinity for the gas. As when compared to other pigments, it will more easily load the gas.
Affinity is defined as the degree to which a substance tends to combine with another and in this case, it is used to describe the degree to which the gas tends to combine with a respiratory pigment.
Respiratory pigment such as Myoglobin has a higher affinity than Haemoglobin to load oxygen.
Learn more about Respiratory Pigments:
https://brainly.com/question/31467554
#SPJ4
What mass of K₂CO₃, in grams, is present in 0.273 L of a 0.998 M solution?
Answers
Answer: Mass of K2CO3 is 37.7g
Explanation: You first need to find the moles of K2CO3 by using the molarity formula.
Molarity = moles/Liters
When you do 0.998 = moles/0.273, you will get 0.272454 moles of K2CO3.
The second step is to use the moles of K2CO3 you found and convert it to grams. As shown in the image. Make sure your final answer has the correct number of significant figures. In the question both of the numbers given have 3 sig figs therefore your final answer also needs to have 3 sig figs.
Calculate the freezing point and the boiling point of each of the following aqueous solutions. (Assume complete dissociation. Assume that water freezes at 0.00°C and boils at 1.86°C 100.000°C. K = 0.51°C Kb = molal molal a. 0.060 m MgCl2 T = °C T = °C b. 0.060 m FeCl3 T = °C To = °C
Answers
The freezing and boiling points of 0.060 m [tex]MgCl_2[/tex] are -0.33°C and 100.09 °C. 0.060 m [tex]FeCl_3[/tex] has the following freezing and boiling points of -0.44°C and 100.12 °C respectively.
Depression in the freezing point and elevation in the boiling point are colligative properties. Colligative properties refer to the properties that are dependent on the concentration of solute in the solution.
Depression in the freezing point is calculated as ΔT = [tex]ik_fm[/tex]
where ΔT is depression in the freezing point
i is the dissociation factor
[tex]k_f[/tex] is the freezing depression factor = 1.86°C kg/mol
m is the molality of the solution
So, depression in 0.060 m [tex]MgCl_2[/tex] is 3*1.86*0.06
( it has 3 as a dissociation factor as it breaks into 1 [tex]Mg^{2+[/tex] and 2 [tex]Cl^-[/tex] ions)
0 - freezing point = 0.33
freezing point = -0.33°C
So, depression in 0.060 m [tex]FeCl_3[/tex] is 4*1.86*0.06
( it has 4 as a dissociation factor as it breaks into 1 [tex]Fe^{3+[/tex] and 3 [tex]Cl^-[/tex] ions)
0 - freezing point = 0.44
freezing point = -0.44°C
Elevation in boiling point is calculated as ΔT = [tex]ik_bm[/tex]
where ΔT is Elevation in boiling point
i is the dissociation factor
[tex]k_b[/tex] is the boiling elevation factor = 0.51°C kg/mol
m is the molality of the solution
So, elevation in 0.060 m [tex]MgCl_2[/tex] is 3*0.51*0.06
( it has 3 as a dissociation factor as it breaks into 1 [tex]Mg^{2+[/tex] and 2 [tex]Cl^-[/tex] ions)
boiling point - 100 = 0.09
boiling point = 100.09 °C
So, elevation in 0.060 m [tex]FeCl_3[/tex] is 4*0.051*0.06
( it has 4 as a dissociation factor as it breaks into 1 [tex]Fe^{3+[/tex] and 3 [tex]Cl^-[/tex] ions)
boiling point - 100 = 0.12
boiling point = 100.12 °C
Learn more about Colligative Properties:
https://brainly.com/question/30575192
#SPJ4
the shattered glass case at the scene of a jewelry store robbery was determined to be made of potash borosilicate glass, which has a density of 2.16 g/ml. a 2.573 g glass fragment was recovered from a suspect's clothing. when the fragment was placed into a graduated cylinder filled with water, 1.14 ml of the water was displaced. calculate the density of the glass fragment.
Answers
The density of the glass fragment is approximately 2.26 g/ml
What is the density of the fragment?To calculate the density of the glass fragment, we can use the formula:
Density = Mass / Volume
First, let's calculate the volume of the glass fragment using the displacement method. The volume of water displaced when the glass fragment was submerged in the graduated cylinder is given as 1.14 ml.
So, the volume of the glass fragment is 1.14 ml.
Next, we can calculate the density of the glass fragment by dividing the mass of the glass fragment by its volume:
Density = Mass / Volume = 2.573 g / 1.14 ml
Density = 2.573 g / 1.14 ml ≈ 2.26 g/ml
Learn more about density here: https://brainly.com/question/6838128
#SPJ1
HELP PLSSSS
What's the molar mass of alumina, Al₂O₂? The atomic weight of aluminum is 26.98 and the atomic weight of oxygen is 16.00.
A. 101.96 g/mol
B. 48.00 g/mol
C. 149.96 g/mol
D. 42.98 g/mol
Answers
Bauxite has a molar mass of 148.96 g/mol. Alumina has an atomic weight of 26.98 and air has an atomic weight of 16.00. As a result, alumina's molar mass equals 42.98 g/mol Plus 26.98 g/mol (= 148.96 g/mol.
The correct answer is :D.
Is aluminum's molar mass 26.98 g mol?One mole of Al atoms possesses a mass in grammes that is numerically comparable to aluminum's atomic mass. According to this regular visual representation, the atomic weight (which was rounded to two decimals places) of Al is 26.98, hence 1 mol of Al atoms weighs 26.98 g.
What does the number 26.98 indicate in terms of aluminium?An aluminium atom possesses a weight od 26.98 amu on average. As a result, one atom of aluminium weighs 26.98 amu. A copper atom possesses an average diameter of 63.55 amu. As a result, a single copper atom weighed 63.55 amu.
To know more about atomic visit:
https://brainly.com/question/30898688
#SPJ1
a gaseous product has a mass of 2.34 g and occupies a volume of 0.854 l. the temperature in the laboratory is 302 k, and the air pressure is 1.04 atm. calculate the molar mass of the gas. (3 points) 44.0 g/mol 86.9 g/mol 65.3 g/mol 22.4 g/mol
Answers
The molar mass of the gas is approximately 65.3 g/mol. The closest answer choice is 65.3 g/mol, so that is the correct answer.
To calculate the molar mass of the gas, we can use the ideal gas law:
PV = nRT
where P is the pressure, V is the volume, n is the number of moles, R is the ideal gas constant, and T is the temperature in Kelvin.
First, we need to calculate the number of moles of the gas using the given information:
n = (PV) / (RT)
n = (1.04 atm * 0.854 L) / (0.0821 L·atm/(mol·K) * 302 K)
n = 0.0361 mol
Next, we can calculate the molar mass of the gas by dividing its mass by the number of moles:
molar mass = mass / number of moles
molar mass = 2.34 g / 0.0361 mol
molar mass = 64.9 g/mol
Therefore, the molar mass of the gas is approximately 65.3 g/mol. The closest answer choice is 65.3 g/mol, so that is the correct answer.
Learn more about molar mass,
https://brainly.com/question/22997914
#SPJ4
This is a machine that converts electrical energy into mechanical energy.
A. Generator
B. Motor
C. Electricity
(why do my questions keep getting deleted?)
Answers
A motor is a machine that converts electrical energy into mechanical energy. It works by using the interaction between a magnetic field and an electric current to generate a rotating force, which can then be used to power a variety of mechanical devices such as pumps, compressors, or machinery.
A generator, on the other hand, is a machine that converts mechanical energy into electrical energy. It works by using a rotating magnetic field to induce an electrical current in a wire coil, which can then be harnessed as electrical power.
Electricity is a form of energy that is carried by the movement of electric charges, and is used to power a wide variety of devices and machines, including motors and generators.
Maltose has a (1→4) α linkage between glucose and ________. xylulose mannose galactose glucose
Answers
Maltose, also referred to as malt, is a disaccharide composed of two alpha-D glucose units. Maltose has a (1→4) α linkage between glucose and glucose.
Maltose, also referred to as malt, is a disaccharide composed of two alpha-D glucose units. An alpha 1,4 glycosidic bond connects the two glucose units. The enzymes maltase and isomaltase break down the molecules of maltose into two glucose molecules in the human small intestinal lining, which are then absorbed by the body. After cellulose, starch is the polysaccharide that is most prevalent in plant cells.
A disaccharide is a type of carbohydrate that is created by joining two units of glucose. Succrose, maltose, and lactose are the three most prevalent types of disaccharide. The other disaccharides are lactulose, trehalose, and cellobiose, which are less well-known.
Visit here to learn more about molecules : https://brainly.com/question/28931982
#SPJ11
when is the residue removal log used? select the correct response. every time you rinse or air dry to remove residue from equipment before using it with organics whenever unloading a chlorine dishwasher at least once per shift
Answers
The residue removal log is used every time you rinse or air dry to remove residue from equipment before using it with organics.
The residue removal log is used whenever unloading a chlorine dishwasher, as it helps track the process of ensuring that equipment is free of residue before using it with organics.
Removal of logging residue negatively affected tree diameter and height, but had no significant effect on the basal area of the subsequent stand (in the mid-term). On the other hand, different methods of mechanical site preparation (bedding, plowing furrows, and trenching) had no effect on tree growth 1 year after planting, but had a significant effect on tree diameter, tree height, and basal area in the mid-term. Bedding treatments could have a significant positive impact on the productivity of the subsequent Scots pine stands, even when planted on sandy, free-draining soils.
Visit here to learn more about soils : https://brainly.com/question/23813511
#SPJ11
prepare a solution of the following concentration: 23 micromoles/liter. measure its absorbance at 400 nm. how will you prepare 1 ml of the assigned solution? below, enter the volume of pnp stock solution you will pipette, and the amount of 0.100 m sodium bicarbonate. answer in microliters.
Answers
To prepare 1 mL of 23 µM/L solution, pipette stock solution and add 17.5 µL of 0.100 M sodium bicarbonate.
To set up an answer of 23 µM/L, first work out the expected measure of solute. For a volume of 1 L, 23 µmol of solute is required. To plan 1 mL of the arrangement, the expected measure of solute is 23 nmol.
Accepting the sub-atomic load of the solute is known, the mass of solute required can be determined. Then, disintegrate the mass of solute expected in a reasonable dissolvable to make a stock arrangement. Weaken this stock arrangement fittingly to set up the ideal grouping of 23 µM/L.
To gauge the absorbance at 400 nm, utilize a spectrophotometer. Set up a clear arrangement utilizing a similar dissolvable and measure the absorbance of this clear at 400 nm. Then, measure the absorbance of the example arrangement and work out the contrast between the two absorbances.
To get ready 1 mL of the relegated arrangement, pipette the necessary volume of the stock arrangement and add 17.5 µL of 0.100 M sodium bicarbonate. This is expecting that sodium bicarbonate is being utilized as a cushion to keep up with the pH of the arrangement.
The specific volume of the stock arrangement required relies upon the convergence of the stock arrangement.
To learn more about stock solution, refer:
https://brainly.com/question/21120916
#SPJ4
explain why the amide nitrogen is much less reactive as a base towards aqueous acids than the alkylamine nitrogen. how does this experiment illustrate this?
Answers
The amide nitrogen is much less reactive as a base towards aqueous acids than the alkylamine nitrogen due to the presence of the carbonyl group adjacent to the nitrogen in the amide.
This carbonyl group withdraws electron density from the nitrogen, making it less basic and less likely to accept a proton from an aqueous acid. In contrast, the alkylamine nitrogen has no such electron-withdrawing group, and thus is more basic and more likely to accept a proton from an aqueous acid.
An experiment that illustrates this difference in reactivity is the acid-base titration of an amide and an alkylamine with hydrochloric acid. The amide would require a stronger acid and a longer titration time to reach its equivalence point, indicating its lower reactivity as a base towards aqueous acids. On the other hand, the alkylamine would require a weaker acid and a shorter titration time to reach its equivalence point, indicating its higher reactivity as a base towards aqueous acids.
Learn more about amide nitrogen here: brainly.com/question/30724866
#SPJ11
mike needs to make 2 liters of a 0.3 m naoh solution for an experiment. he has a 1.2 m stock solution. how much stock solution must be diluted to reach his desired 2 liter, 0.3 m final solution?
Answers
Mike needs to dilute 0.5 liters of the 1.2 M NaOH stock solutions to reach his desired 2-liter, 0.3 M final solution.
To make a 2 liter, of 0.3 M NaOH solution, Mike needs to dilute his 1.2 M stock solution. The calculation can be done using the formula:
C1V1 = C2V2
where C1 is the concentration of the stock solution, V1 is the volume of the stock solution to be diluted, C2 is the desired concentration of the final solution, and V2 is the total volume of the final solution.
Rearranging the formula to solve for V1, we get:
V1 = (C2V2) / C1
Plugging in the values, we get:
V1 = (0.3 M x 2 L) / 1.2 M
V1 = 0.5 L
Therefore, Mike needs to dilute 0.5 liters of his 1.2 M stock solution with water to make a 2 liter, 0.3 M NaOH solution.
Learn more about stock solutions at https://brainly.com/question/30970534
#SPJ11
Mike needs to dilute 0.5 liters of his 1.2 M stock solution to make 2 liters of a 0.3 M NaOH solution for his experiment.
To make a 2 liter, 0.3 M NaOH solution, Mike will need to dilute his 1.2 M stock solution.
The equation for dilution is: C1V1 = C2V2, where C1 is the initial concentration, V1 is the initial volume, C2 is the final concentration, and V2 is the final volume.
We can rearrange this equation to solve for V1, which is the volume of stock solution that Mike needs to dilute.
C1V1 = C2V2
V1 = (C2V2)/C1
In this case, C1 = 1.2 M, C2 = 0.3 M, and V2 = 2 liters.
V1 = (0.3 M * 2 liters)/1.2 M = 0.5 liters
So Mike needs to dilute 0.5 liters of his 1.2 M stock solution to make 2 liters of a 0.3 M NaOH solution for his experiment.
Learn more about solution here:
https://brainly.com/question/20067151
#SPJ11
ibuprofen has the following mass percent composition: c 75.69 % , h 8.80 % , o 15.51 % . what is the empirical formula of ibuprofen?
Answers
Rounding these values to the nearest whole number, we get the empirical formula of ibuprofen as C6H9O.
To determine the empirical formula of ibuprofen, we need to convert the mass percent composition into mole ratios. This can be done by assuming that we have 100 grams of ibuprofen, and calculating the number of moles of each element present in that sample.
Starting with carbon, we have 75.69 grams of carbon in our sample, which corresponds to 6.30 moles (using the atomic weight of carbon). Similarly, we have 8.80 grams of hydrogen, which corresponds to 8.74 moles, and 15.51 grams of oxygen, which corresponds to 0.97 moles.
To get the simplest whole number ratio of these elements, we divide each mole value by the smallest one (0.97):
- Carbon: 6.30 / 0.97 = 6.49
- Hydrogen: 8.74 / 0.97 = 9.00
- Oxygen: 0.97 / 0.97 = 1.00
This means that the molecular formula of ibuprofen could be a multiple of this empirical formula (e.g. C12H18O2), but we would need additional information (such as the molecular weight) to determine that.
To learn more about : ibuprofen
https://brainly.com/question/15179513
#SPJ11
sodium hydroxide can react with the solvent, ethanol, in this experiment instead of making the enolate. why is this not a problem?
Answers
There is a possibility that sodium hydroxide may react with ethanol to form sodium ethoxide and water instead of forming the enolate.
What is ethanol ?Ethanol, also known as ethyl alcohol or grain alcohol, is a clear, colorless liquid with a slight odor and a burning taste. It is the most common type of alcohol found in alcoholic beverages, and is also used as a solvent, fuel, and antiseptic.
Chemically, ethanol is a simple alcohol with the molecular formula C2H5OH. It is produced through the fermentation of sugars by yeast or other microorganisms, and is also commonly synthesized from ethylene through the process of hydration.
Ethanol has a wide range of uses in industry and everyday life. It is used as a fuel additive to increase octane levels in gasoline, as a solvent in perfumes, cosmetics, and pharmaceuticals, and as a disinfectant for medical instruments and surfaces. In addition, ethanol is a common recreational drug, and is consumed in the form of beer, wine, and spirits.
To know more about Ethanol visit :
https://brainly.com/question/25002448
#SPJ1
converting numbers to scientific notation?
Answers
Scientific notation is a way of expressing very large or very small numbers using powers of 10.
How to convert numbers to scientific notation?The general form of a number in scientific notation is:
a × 10^b
where;
a is a decimal number between 1 and 10 (inclusive), and b is an integer representing the power of 10.To convert a number to scientific notation, follow these steps:
Identify the decimal point in the number. If the number is an integer, assume the decimal point is at the end of the number (e.g., 100 is the same as 100.0).
Move the decimal point to the right or left so that only one non-zero digit remains on the left side of the decimal point.
Count the number of places the decimal point moved. This will be the value of "b" in the scientific notation.
The remaining number on the left side of the decimal point is "a" in the scientific notation.
Write the number in the form "a × 10^b".
Here's an example:
Number: 2450
Identify the decimal point: 2450.
Move the decimal point to the left after the first digit: 2.450.
Count the number of places the decimal point moved (in this case, 3 places to the left): b = 3.
The remaining number on the left side of the decimal point is 2.45: a = 2.45.
Write the number in scientific notation: 2.45 × 10^3.
Learn more about scientific notation here: https://brainly.com/question/5756316
#SPJ1
I need to the the answers for the boxes
Answers
In the case of the equilibrium reaction provided, if the concentration of Co(H2O)2+ is increased, the reaction will shift to the right in order to consume the excess Co(H2O)2+ and produce more CoCl2 and H2O.
Conversely, if the concentration of CoCl2 is increased, the reaction will shift to the left in order to consume the excess CoCl2 and produce more Co(H2O)2+ and Cl- ions.
How would a change in concentration affect equilibrium?According to Le Chatelier's principle, a change in concentration of one or more of the reactants or products of a chemical reaction at equilibrium will cause a shift in the equilibrium position to counteract the change and re-establish equilibrium.
Specifically, if the concentration of one of the reactants is increased, the reaction will shift in the direction that consumes that reactant in order to restore equilibrium.
Learn more about chemical equilibrium at: https://brainly.com/question/18849238
#SPJ1
the salt of codeine, codeine bromide (c18h21o3nh br- ) has analgesic and antitussive properties. calculate the ph of a 0.324 m codeine bromide solution. pkb of c18h21o3n is 7.95. keep your answer to 3 decimal places.
Answers
The pH of a 0.324 M codeine bromide solution is 9.743.
To calculate the pH of a solution of codeine bromide, we need to determine the concentration of hydroxide ions (OH⁻) present in the solution. Codeine bromide is a salt, so it dissociates in water to produce codeine cations (C₁₈H₂₁O₃N⁺) and bromide anions (Br⁻).
The codeine cation can act as a weak base and react with water to produce hydroxide ions (OH⁻) and the conjugate acid of codeine. Since the pKb of codeine is known (7.95), we can use the following equation to calculate the concentration of hydroxide ions:
Kb = Kw / Ka = [OH⁻]² / [C₁₈H₂₁O₃N⁺][OH-] = √(Kb*[C₁₈H₂₁O₃N⁺]) = sqrt(10⁻¹⁴ / 10^(7.95)) * 0.324 M = 1.657 x 10⁻⁴ MpH = 14 - pOH = 14 + log([H₃O⁺]) = 14 - log([OH⁻]) = 9.743Therefore, the pH of a 0.324 M codeine bromide solution is 9.743.
To learn more about pH of solution, here
https://brainly.com/question/30934747
#SPJ4
A liquid typically boils at 200 degrees C. If an insoluble salt is added to the liquid, its new boiling point will be:
a) greater than 200 degrees C
b) less than 200 degrees C
c) 200 degrees C
d) not enough information is provided to answer the question
Answers
Answer:
Explanation:
the addition of the soluable salt will cause the boiling point to be higher
a) greater than 200 degrees C. if an insoluble salt is added to a liquid that typically boils at 200 degrees C, its new boiling point will be greater than 200 degrees C.
When an insoluble salt is added to a liquid, it causes a change in the vapor pressure of the liquid, which in turn affects the boiling point of the liquid. The addition of an insoluble salt to a liquid raises its boiling point.
This is because the presence of the solute in the liquid reduces the vapor pressure of the liquid, making it more difficult for the liquid to boil. The boiling point of the liquid increases until the vapor pressure of the liquid once again matches the external pressure, at which point the liquid will boil.
Learn more about insoluble here:
https://brainly.com/question/18799126
#SPJ11
In the SN1 reaction of 2-chloro-2-methylpropane with water at different temperatures, the following reaction rate constants were obtained: 17°C, 0. 0052 s-1; 30°C, 0. 0202 s-1; 42°C, 0. 0608 s-1. Calculate the half-life of this reaction at 36°C. In seconds
Answers
The half-life of the reaction at 36°C is 19.2 seconds.
We can use the following equation to determine the half-life of the reaction at 36°C;
[tex]t_{1/2}[/tex] = ln(2) / k
where [tex]t_{1/2}[/tex] is the half-life of the reaction and k is the reaction rate constant at the given temperature.
First, we need to find the reaction rate constant at 36°C. We can use the two rate constants given for 30°C and 42°C and the Arrhenius equation;
ln(k₂/k₁) = (-Ea/R) × (1/T₂ - 1/T₁)
where k₁ and k₂ are the rate constants at temperatures T₁ and T₂, Ea will be the activation energy, R is gas constant, and T is temperature in Kelvin.
We can choose 30°C (303 K) as T₁ and 42°C (315 K) as T₂, and solve for ln(k₂/k₁) to get;
ln(k₂/k₁) = (-Ea/R) × (1/T₂ - 1/T₁)
ln(0.0608/0.0202) = (-Ea/8.314 J/(mol×K)) × (1/315 K - 1/303 K)
Ea ≈ 52.7 kJ/mol
Next, we can use the Arrhenius equation to find the rate constant at 36°C (309 K);
k = A × exp(-Ea/RT)
k = 0.0202 s⁻¹ × exp(-52.7 kJ/mol / (8.314 J/(mol×K) × 309 K))
k ≈ 0.036 s⁻¹
Finally, we can use the half-life equation with this rate constant to find the half-life at 36°C;
[tex]t_{1/2}[/tex]= ln(2) / k
[tex]t_{1/2}[/tex] = ln(2) / 0.036 s⁻¹
[tex]t_{1/2}[/tex] ≈ 19.2 s
To know more about half-life here
https://brainly.com/question/24710827
#SPJ4
How much 8.0 M stock solution is required to prepare 100.0 mL of 2.5 M
solution?
Answers
V1=M2×V2/M1
Plug in the known values and solve for your unknown:
V1=0.50M×100.0mL2.5M
Therefore,
V1=20.mL
How is a 1M stock solution made?The term "molarity" (M) refers to the quantity of solute in moles per litre of solution. A clean 1-L volumetric flask should be halfway filled with distilled or deionized water to create a 1 M solution. Slowly add 1 formula weight of the chemical to the flask. Allow the compound to completely dissolve, gently turning the flask as needed.
Consider making 50 millilitres of a 1.0 M solution from a 2.0 M stock solution, as an example. Calculating the volume of stock solution needed is the first thing you should do. Pour 25 ml of the stock solution into a 50 ml volumetric flask to create your solution.
learn more about molarity
https://brainly.com/question/30404105
#SPJ1